Oil and water don't mix, right? That's why you shake up your salad dressing; to temporarily force them together. Why does adding a little dish detergent (soap) help to remove the grease from dirty dishes better than water alone? Why does dry cleaning lift stains that soap and water won't? The answer has to do with the chemical properties of the solvents we use, and the chemical properties of the things we are trying to dissolve (the solutes). We'll come back to these examples later.

Chemical Bonds: Atoms seek more stable states. The structure of an atom is similar to that of the solar system. The large protons (with a positive charge) and neutrons (with no charge) are found at the nucleus or center. The tiny electrons (with negative charges) circle rapidly in orbits around the nucleus, forming electron shells at different distances, much like the planets and other objects that circle the sun. Atoms of each element have varying numbers of electrons in their outermost shells. Atoms become more stable when their outermost electron shells are emptied out or filled up. One way they can achieve this goal is for two atoms to share one or more electrons between them so that each of them can fill or empty that outermost shell. But they can only share the electron(s) if they stay close to each other, and this is called a covalent bond. In other situations, one atom can become more stable by losing electrons and the other can become more stable by gaining them. The atom that gained an electron (remember that electrons have a negative charge) becomes negatively charged (-1) while the atom that lost an electron becomes positively charged (+1). Here's a little joke to help you remember...

When an atom loses an electron, its net charge goes from 0 (neutral) to +1 (positive)
The formation of an ionic bond is a redox reaction. One atom loses electrons (oxidation) while the other one gains electrons (reduction). Atoms that carry a charge, either positive or negative, are called ions and, because opposites attract, they can form an ionic bond. Ionic and covalent bonds are the most important in all of chemistry. Here's a little joke to help you remember...

With ionic bonds, atoms give or take electrons. With covalent bonds, they have to share them.


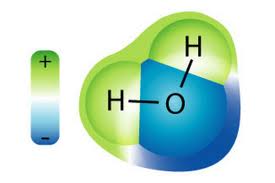
Now think about a magnet. Magnets have both a positive (+) pole and a negative (-) pole. So do batteries. So does the Earth. When things are different at each end, we call them polar. Some molecules have positive and negative ends too, and when they do, we call them polar. If they don't, we call them non-polar. Things that are polar can attract and repel each other (opposite charges attract, alike charges repel). The two magnets in the image above will attract because their opposite poles are near. Reverse one of them and they will repel each other.
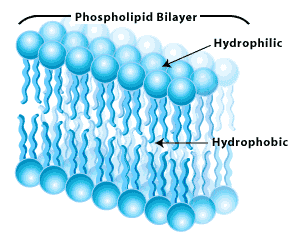

So why do soaps and detergents clean our dishes and our clothes? Soaps are chemically similar to cell membranes. When soap is added to water, it forms structures called "micelles." The heads of the soap micelles are polar and the tails, which face inward to retreat from the polar water, are non-polar. When a soap micelle encounters oil or grease, these non-polar materials are forced to the inside of the micelle to get away from the polar water and polar heads of the micelle, where they are trapped. When the soapy water is rinsed away, the trapped grease and oil is washed away with it.
Mini-Experiment 1: Pour some water into a shallow bowl. Now take a length of thread or a long hair and lay it on top of the water in a closed loop. Put a few drops of vegetable oil inside the loop of thread and gently stir the oil. Now add some dish detergent outside the loop of string and gently stir it into the water. Remove the thread and watch what happens.
Mini-Experiment 2: Here's a dramatic experiment you can do with food coloring, dish soap, and milk. Watch the video to see how it will look. Why does this work? Hint: milk contains fats, and soap repels fats. The food coloring is carried along in the milk as it retreats from the fats.